Performance data for monitored care decisions.
Mindray performance documentation is organized for clinical governance, biomedical engineering, informatics, and procurement reviewers who need to test claims before they become purchasing assumptions.

Evidence areas reviewed before implementation
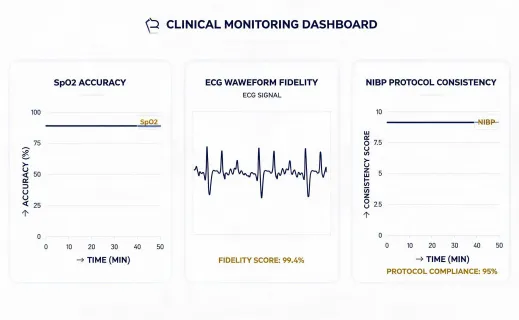
Clinical performance data becomes useful only when it is attached to a care workflow. For Mindray patient monitoring, evaluation packets should identify ECG waveform export behavior, SpO2 accuracy assumptions, NIBP measurement protocols, alarm priority behavior, event storage, and central station routing. For respiratory and anesthesia platforms, the evidence set should describe gas module verification, ventilation modes, transport battery behavior, service intervals, and alarm escalation. For home health programs, the record should include caregiver training, pairing instructions, adherence data, HIPAA-safe communications, and device replacement procedures.
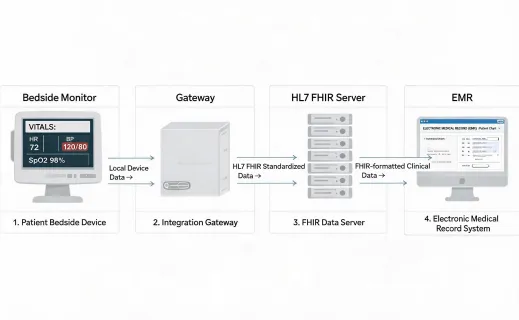
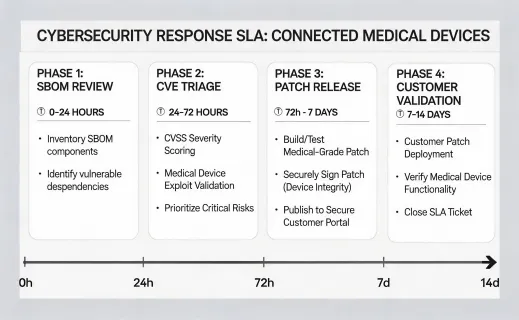
The performance-data page also gives IT and security teams a defined review path. Connected medical devices should be evaluated for SBOM access, MDS2 documentation, encryption in transit, role-based access, certificate management, vulnerability disclosure, and patch validation. Informatics teams need a separate interoperability matrix that names HL7 messages, FHIR resources, DICOM touchpoints, device identifiers, and the expected cadence of observations. When these topics are separated from marketing copy, clinical committees can compare claims against their own operating controls.
Mindray buyers should request the specific packet that matches their deployment: ICU and step-down monitoring, anesthesia and respiratory continuity, remote patient monitoring, or home rehabilitation. Each packet can include IFU references, cybersecurity notes, integration assumptions, service coverage, training plans, warranty limits, and post-market surveillance summaries.